Probing cell membrane penetration mechanism of enveloped viruses (CoV, IFV, HIV, Ebola, etc.) using nano-FTIR and spectroscopic nanoscale imaging.
Researchers from the University of Georgia and Georgia State University utilized neaSCOPE for investigating how an individual enveloped virus penetrates the membrane of a host cell at the nanoscale. Viruses can enter the host cell differently. The most common process for enveloped viruses involves a complex fusion of the virus into the cell membrane. Here, researchers performed a direct quantitative investigation of the virus-cell membrane fusion process at a single virus level by employing IR nanoscale imaging and nano-FTIR spectroscopy. Such comprehensive studies of the cell membrane penetration behavior by viruses provide important information for antiviral therapies and vaccines development against many human infections such as Influenza, HIV, Coronavirus, Ebola, and others. This work has been recently published in PLOS ONE.
Enveloped viruses have their genome surrounded by a phospholipid envelope while being between host cells. The cell membrane penetration by an enveloped virus is a key step in the process of cell infection. Specifically, it is important to understand how the enveloped virus interacts with the host cell receptors and what structural changes are occurring in the envelope itself during the membranes fusion. Numerous studies have established a recognized model for the fusion mechanism between the target and the viral membranes. This model assumes that pores can be formed only when the target and the viral membrane undergo membrane fusion to mediate virus-cell membrane permeation. However, recent observations indicate that the target and viral membranes rupture before the fusion occurs. In addition, studies on adenovirus proteins and host cells have shown that host cell membranes can be destroyed upon the virus entry without the membranes fusion. On the other hand, the virus envelope and the target host cell membrane have different chemical compositions or structures. Therefore, the requirements for forming pores in each membrane are different, so the targeted host or virus membrane rupture may also be induced independently. To sum up, there is still a controversy about the mechanism of virus-cell membrane penetration behavior. This is because it is highly nontrivial to provide a comprehensive direct and quantitative observation for an individual fusion process, since due to small size of the viral particles, it requires chemically specific imaging with nanoscale spatial resolution. Clarification of such complex collective fusion mechanism of merging between a single virus and a host cell can provide beneficial information for the design of antiviral compounds.
Changes in the chemical and structural composition of viral and host cell membranes caused by the viral infection process at the molecular level can be detected by infrared spectroscopy. However, the characteristic size of viruses, lipid envelopes and surface glycoproteins mediating the fusion process is much smaller than the diffraction limit for infrared light, which prevents single particle studies. Therefore, it is important to find a tool that can provide nanoscale spatial resolution while detecting mechanical & chemical properties (e.g. molecule specific infrared absorption), as well as environmental impacts, so that the virus and cell membranes fusion process can be fully analyzed.
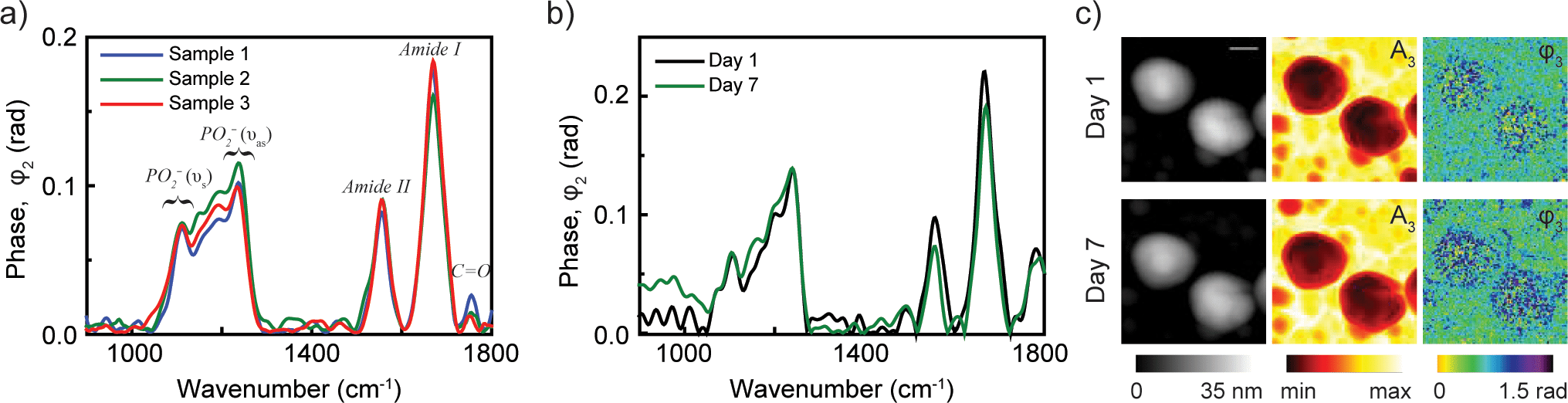
(a) nano-FTIR spectra of influenza virus particles from three different samples at neutral pH demonstrate the reproducibility of nanoscale analysis. (b) Spectra from virus particles taken on day 1 and day 7 shows the stability of viral particles. (c) Topography, near-field amplitude (A3) and phase (φ3) images representing reflectivity and absorption of two influenza virus particles on day 1 and day 7. Scale bar 100 nm. From PLOS ONE
Researchers at Yohannes Abate’s and Ming Luo’s groups used spectroscopic IR nanoimaging provided by neaSCOPE to study chemical and structural changes that occur prior to membrane fusion in a single archetypal enveloped influenza virus X31 in different pH environments. With fully quantitative data provided by nano-FTIR, the group was also able to quantitatively evaluate the effectiveness of the antiviral compound (Compound 136) in preventing the disruption of viral membrane – a novel mechanism for inhibiting the virus from entering the cell.
With IR nanoimaging and nano-FTIR spectroscopy, neaspec microscopes provide a unique tool for nanoscale analysis of basic mechanisms underlying the functionality of virus and cell membranes, which can significantly boost progress in fundamental viral research and treatment development.